Inflammatory bowel disease (IBD) is characterized by chronic and recurring inflammation and activation of the immune response in the gastrointestinal tract. The two most common forms of IBD are ulcerative colitis (UC), distinguished by inflammatory mucosal lesions in the colon, and Crohn’s disease (CD), defined by transmural lesions that can develop anywhere in the digestive tract. IBD patients often experience intestinal bleeding, obstruction, and fistula formation, and up to 75% of patients with CD and 25% of those with UC will eventually require surgery.
IBD affects 1.4 million persons in the US, and the incidence rates for CD and UC have increased to 19.2 and 20.2 per 100,000 persons per year, respectively. As such, IBD now accounts for more than 700,000 physician visits, 100,000 hospitalizations, and 119,000 disability cases each year, and its annual health care costs are expected to increase from $1.7 billion in 2011 to $3.2 billion by 2017.
IBD has no cure and generally requires lifelong medical care. Conventional and more recently developed biological therapeutic agents, however, have very limited efficacy. The best treatments available typically fail to induce remission and, after 2-5 years of treatment, one fifth of patients will still require surgery. Therefore, there is a pressing need to develop better therapeutic agents to control IBD and induce lasting deep IBD remission.
Preclinical studies:
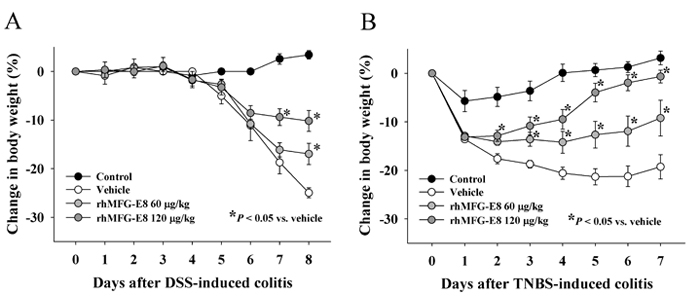
rhMFG-E8 prevents weight loss in IBD. Treatment with rhMFG-E8 significantly decreased the weight-loss caused by (A) DSS- and (B) TNBS-induced colitis, two well-established experimental models of IBD. Vehicle (saline) or rhMFG-E8 (60 or 120 µg per kg of body weight) was administered daily subcutaneously, starting at day 2 in the DSS model and at day 1 in the TNBS model. Data are presented as mean ± SE (n=5/group) and compared using one-way ANOVA with the Student-Newman-Keuls test; *P < 0.05 vs. vehicle.
